Greek Philosophers:
Democritus was an ancient Greek philosopher and lived from 460 B.C. to 370 B.C. He was one of the first scientists to discover the atom. He stated that everything is made up of atoms. He also believes that there are a numerous amount of atoms that different from size and shape. He only thought that an atom doesn't have any charges or a nucleus. In his theory he also stated that atoms are solid.
John Dalton:
John Dalton was born on September 6, 1766 in Cumbria, United Kingdom. He was an English chemist and had his own model of the atom. He stated that atoms are too small to be seen by the human eye. He also acknowledged that compounds are formed by the joining of atoms of two or more elements. His model is known as Dalton's Atomic Model.
Joseph John Thomson
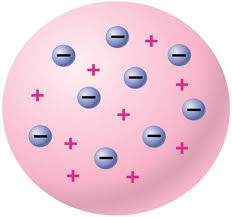
Joseph John Thomson was born on December 18, 1856 in Cheetham Hill, it is a suburb in Manchester. In 1897, he provided the first hint that atoms were made up of even smaller particles. He stated that atoms are made up of positively and negatively charged particles. He went to Trinity Collage at Cambridge and majored in mathematics and physics. His model is know as a plum pudding.
Ernest Rutherford
Ernest Rutherford was born on August 30,1871 in Spring Grove, New Zealand. In 1908, he discovered the nucleus. His model had a nucleus with protons inside and electron surrounding the nucleus. He was a former student in JJ Thomson's class and proved that his model was incorrect. He stated that's atoms have a small dense positively charged center called the nucleus. His model is known as Rutherford's Model.
Modern Atomic Model
The modern atomic model is also known as the electron cloud. It was discovered by Erwin Schrodinger in 1925. It has a tiny nucleus with protons and neutrons inside the nucleus. On the outside contains a lot of electrons. It is a neutral atom because of the electrons.
Period Table Facts1) There are 118 elements known as for today.
2) The difference between the modern periodic table and Mendeleev's periodic table is that Mendeleev's table arranged the elements in order of increasing atomic weight while the modern table orders the elements by increasing atomic number. 3) The two sections are called groups and periods. 4) It contains solids, liquids, and gasses. 5) It also contains metals, nonmetals, and metalloids. 6) Helium is on the far left because it is part of group zero. 7) Moseley found elements that were out of order in Mendeleev's table. 8) It is called "periodic" because elements are lined up in cycles or periods. 9) Mosely made a periodic table based on atomic number, not atomic mass. 10) Some abbreviations don't go along with the real word. |